Review Article - (2022) Volume 13, Issue 8
Abstract
The demanding sector of efficient brain delivery has great vision of scientific interest, resulting in the invention and patenting of numerous novel methods. Therefore in this article, we discuss recent remarkable developments in the use of nanotechnology for medication delivery. Central Nervous System (CNS) conditions, specifically neurodegenerative disorders, are a serious public health issue that researchers address if individuals are to be saved. To promote treatment efficacy while minimizing negative impacts, diverse polymers have been incorporated in the preparation of Nanoparticles for drug discovery investigations. Pharmaceutical nanotechnology-based systems, preparation techniques, uses, benefits, and drawbacks and the most significant developments in the field of nanotechnology as a pharmaceutical means of delivering are highlighted in this review article.
Keywords
Nanoformulations, Nanoparticles, Targeting brain drug delivery
Introduction
Nanotechnology is the study of the science of nanoparticles. It is the manipulation and application of matter on a microscopic level. Atoms and molecules respond differently at this magnitude that provides a range of unique and unexpected effects. Nanostructures and nonmaterial have progressed substantially in a multi tude of new fields in recent decades. It leads to the emergence of goods, particularly for therapeutic applications, where traditional methodologies have hit their limits. Pharmaceutical nanoparticles are submicron-sized solid drug carriers which were either biodegradable or non-biodegradable. Nanoparticle is a word that applies to both nanospheres and nanocapsules. Nanospheres are a matrix system in which the drug is scattered within a polymer matrix, whereas nanocapsules are reservoir systems where the drug is held in the internal cavity of the polymeric membrane. Polymeric nanoparticles can be made of natural polymers such as gelatin, pectin, sodium alginate, or synthetic polymers such as Poly Lactic-co-Glycolic Acid (PLGA), Poly Butyl Acrylate (PBA), and Polylactic Acid (PLA). Drugs, proteins, and DNA are distributed adequately to targeting cells and organs through polymeric nanoparticles. Their molecular size helps in effective absorption and stability across cellular membrane in the bloodstream. Nanotechnology has broadly knocked distinguishable fields of knowledge and has attained respectable progresses in field of medication and medicine delivery (Taghipour YD, et al., 2018). This aids in the treatment of chronic diseases by targeting the site of action and targeted release of API very precisely. The challenges faced by scientist to deliver drug into the brain is achieved by nano formulation as their size range between 10-100 nm (Patra JK, et al., 2018).
Literature Review
Solid Lipid Nanoparticles (SLN)
Solid Lipid Nanoparticles (SLN) are colloidal carriers composed of a biomaterial lipid matrix that are solid at body temperature and size ranging between 100-400 nanometers. Controlled drug release, targeted distribution, superior drug stability, large-scale synthesis, and simplicity of sterilizing are few of the benefits of SLN. A newer type of SLN known as Nanostructured Lipid Carriers (NLC), which are composed of a solid lipid and a modest amount of liquid lipid (oil) and maintain the solid state at both room and body temperature, have recently been popular for brain targeting (Lin LN, et al., 2010; Rangasamy M, 2011).
Nanospheres
Nanospheres, also known as matrix-type nanodevices, they are solid polymeric NPs composed of circular polymeric matrices that have been widely investigated as carriers of medicinal compounds. The biological active agents are evenly dispersed throughout the polymeric core, and the loaded active ingredient is released into the environment by diffusion. Drugs can be adsorbed on the nanospheres’ surface (González-Nieto D, et al., 2020). These nanospheres have the potential to integrate hydrophobic medicines at concentrations larger than their intrinsic water solubility, resulting in prolonged and regulated drug release patterns and high loading capacity for poorly water-soluble pharmaceuticals. The time it takes for the medicine to be released from nanospheres is determined by the polymer matrix’s composition and capacity to absorb fluid (Tian X, et al., 2021). Drug loading and the size of the nanospheres impact efficient drug release from these nanostructures. In general, in release trials, a significant burst impact and quicker release rate are found at increased drug loading. Furthermore, bigger nanospheres have a lower initial burst effect and a longer sustained release than smaller nanospheres. Drug release is mediated by a variety of methods, including hydration-induced degradation of the polymer matrix or swelling, enzymatic or hydrolytic breakage of polymer boundaries, and transport of the physically encapsulated drug (Johanson CE, et al., 2005; Johanson CE, 1980).
Nanogels
Nanogels are nanoparticles made up of a hydrogel that is strongly cross-linked with hydrophilic polymer chains, either mechanically or chemically (Biddlestone-Thorpe L, et al., 2012). The presence of hydrophilic functional groups in nanogels allows them to store a large quantity of water. Nanogels, among other novel drug delivery vehicles, are particularly promising for use in a range of methods, diagnostics, macromolecules, and other applications (Laquintana V, et al., 2009). Nanogels, which are a few nanometers in size, are far better at carrying physiologically active molecules than hydrogels. Chemical modification of nanogels aids in the incorporation of targeting ligands for targeted drug delivery and the inhibition of particular site drug release by external stimuli. When compared to physically cross-linked nanogels, chemically cross-linked nanogels offer a wide range of applications and shows stable behavior. Nanogel production can significantly speed up bioactive molecule encapsulation, making them potentially effective nanomedical carriers. Moreover, they have a greater surface area and drug loading capacity, making them appealing for in vivoapplications (Ohgaki H and Kleihues P, 2007).
Different delivery approach to brain
Considering pharmaceutical molecules passage through the BBB and BCSF, drug delivery to the brain for the treatment of multiple diseases and disorders is extremely complicated and challenging. To overcome such issues and obstacles, brain targeting techniques such as invasive, non-invasive, olfactory drug administration, ophthalmic drug delivery, and focused ultrasonography methods are utilized. They enable easy penetration of therapeutic agents through the BBB and CSF, hence boosting therapeutic efficacy (Burgess A and Hynynen K, 2013).
Brain: The cerebral hemispheres and the brain stem make up the brain. The brain stem is split into three sections: The hindbrain, the midbrain, and the diencephalon. The spinal cord continues in the hindbrain. It regulates blood pressure and respiration. The midbrain comprises groupings of neurons that influence the activity of neurons in the brain by using a specialized type of chemical messenger. The thalamus and the hypothalamus are two components of the diencephalon: The thalamus transfers sensory information to the cerebral cortex, which then transmits information to the thalamus. The hypothalamus modulates mood and also controls processes such as eating and drinking (Komarova Y and Malik AB, 2010; Ghersi‐Egea JF and Strazielle N, 2001).
Barriers of brain: Different processes, including as passive diffusion, transporters, and transcytosis, are part of the transcellular route (Rautio J, et al., 2008; Rodriguez A, et al., 2015) Blood Brain Tumor Barrier (BBTB). Brain cancerous cells mimic the BBB in their early stages because of their rapid proliferation and mobility. The Blood Brain Barrier (BBB) is formed by basal membrane and brain cells namely pericytes and astrocytes (Wallez Y and Huber P, 2008; Rao KS, et al., 2008). The capillaries in brain are formed by the polarized endothelial cells, neurons, tight junctions, where the endothelial cells are surrounded by astrocytes and pericytes which makes the BBB highly impermeable. The two main barriers that restrict the entry of toxic compounds into brain are Blood Brain Barrier (BBB), Blood Cerebrospinal Fluid Barrier (BCSFB), Blood Brain Tumor Barrier and Arachnoid barrier. As a result, the BBB and the BBTB collectively constitute a significant barrier for drug delivery to treat neurological disorders (Taddei A, et al., 2008; Pober JS and Sessa WC, 2007) Brain Cerebrospinal Fluid Barrier (BCSFB) In cerebral ventricles, the choroid plexus is a vascular tissue. As a byproduct, new blood vessels (brain tumor capillary) develop the Blood Brain Tumor Barrier (BBTB), which is separate from the BBB. The BBB is a diffusion barrier preventing neurotoxic chemicals from entering brain cells and ensures brain homeostasis. By the linkage of endothelial cells and interendothelial cells the water soluble substance are restricted from entering into brain. Between the circulating blood and the cerebrospinal region, BCSFB acts as a barrier (Erdő F, et al., 2018). The BBB, on the other hand, is sealed by tight junctions and lacks permanent fenestration (Wallez Y and Huber P, 2008). They play an important role in the transport of metabolites and foreign substances between the blood and the Cerebrospinal Fluid (CSF) (Bahadur S, et al., 2020). Particles pass through the BBB by passive transport through the paracellular route between neighboring cells, utilizing concentration gradients. The Multidrug Resistance-related Proteins (MRPs) and multidrug transporters P-glycoprotein (PgP) are the two main transporters of ATP Binding Cassette. It is essential for the healthy functioning of the brain. The BCSF barrier’s neuroprotective function relies heavily on SLC transporters (Teleanu DM, et al., 2018). The Multidrug Resistance-related Proteins protect the BCSFB against metal ions. The majority of the transporters are ATP-Binding Cassette (ABC) transporter and the Solute Carrier (SLC) super-families. In glioblastomas, BBTB penetration is considerable in the bulk of the tumor, but it is limited or non-existent in the peripheral area. The BCSFB includes multiple gap junctions and pinocytosis vesicles which serve as a protein filter. The BBB is disrupted when cancerous cells reach a particular threshold (Mehdipour AR and Hamidi M, 2009). In the apical side of the Cerebral Palsy (CP) present the low density lipophilic receptor-related proteins which aids in the clearance of peptides form the CSF (Radocha J, et al., 2021; Erdő F, et al., 2018). SLC29A4 is an organic cation transporter that eliminates serotonin, dopamine, and histamine from the CSF since they might involve in the termination of neurotransmitter signaling. The movement of substance into BBB is precisely regulated by protein complexes called interendothelial junctions. The functional unit of CP consists of a capillary surrounded by a layer of modified ependymal epithelium. Occludin, claudins, junctional adhesion molecules are some of the proteins in tight junction (Figure 1).
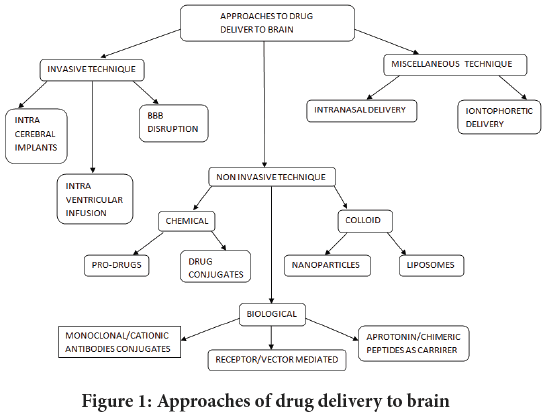
Figure 1:Approaches of drug delivery to brainhes of drug delivery to brain
Invasive technique
Intra cerebral implants: This strategy uses delivering therapeutics directly into the brain parenchymal zone using one of 3 techniques direct infusion viaintrathecal catheter, controlled release matrices, or microencapsulated chemicals. Diffusion is the fundamental mechanism. This type of drug delivery is essential in the treatment of several CNS illnesses, including as Parkinson’s disease (Slavc I, et al., 2018).
Intra cerebral ventricular infusion: At only 1-2 mm from the surface, the estimated quantity of a drug in the brain is only 1%-2% of the CSF concentration. Drugs are delivered effectively to the brain’s surface via an Intracerebroventricular injection (ICV), but they are not carried efficiently to the subarachnoid space. If the medications target receptors are localized near the ependymal surface of the brain, the pharmaco-logical effects can be noticed following Intra-ventricular delivery. The disadvantage is that drug diffusion is relatively poor in the parenchyma, and unless the target is near to the ventricles, this approach of brain focused drug administration is ineffective (Ningaraj NS, et al., 2002).
BBB disruption: This technology was developed in 1989 and was first per- formed on individuals. When the BBB is disrupted, the tight connection between the endothelial cells of the brain capillaries leaks, letting compon- ents in the blood to enter the brain cells. According to this procedure, the barrier is temporarily disrupted by injecting mannitol sugar solution into cervical arteries, resulting in a greater sugar concentration in brain capil- laries and fluid flows out of endothelial cells, weakening them and bypass- ing the tight junction (Avhad PS, et al., 2015).
Noninvasive technique
Chemical techniques:
Prodrug: Prodrugs are defined as drugs that must undergo chemical con- version by metabolic activities to become an active pharmacological agent. The prodrug strategy is used to make a drug more lipophilic by chemical modification. Morphine, for example, never enters the CNS on its own. Morphine may readily cross the BBB after latentiation viaacetylation of both hydrophilic groups and reach an optimum dose in the brain (Dixit N, et al., 2007).
Drug conjugates: The volume of distibution is often increased when molecules are lipidized. Lipophilic addition and modification of hydrophilic substances are two chemical techniques. One of the examples is N-methylpyrimidium 2 carbaldoxime chloride (Dixit N, et al., 2007)
• Analogs of several opioid drugs that have been glycosylated.
• Increase access with antioxidants and pyrrolopyrimidines.
• Ganciclovir enhance transport via hydroxymethyl group+1-methyl 1,4-dihydronicotinate.
Use of fatty acids such as N-docosahexaenoyl (DHA) increases absorption of tiny medicines.
Biological techniques:
Monoclonal antibodies conjugate: The study of the physiological and anatomical details of BBB transportation is the core of biological approaches to CNS medication delivery. Cross-linking of a medication with antibodies is an effective method among the multiple possibilities. Other biological targeting study utilizes ligands such as sugar or lectins, which may be directed to particular receptors on cell surfaces. The antigen on or inside the target tissues is directed by the antibody-drug conjugate. Due to their high affinity and specificity of their ligands, antibodies are particularly well designed for targeting BBB receptor-mediated transcytosis systems (Ghersi-Egea JF and Strazielle N, 2002).
Miscellaneous technique:
Intra nasal route: Nasal route of delivery is also termed as olfactory drug delivery. The nasal tract is a mechanism for delivering active pharmaceutical substances for local, systemic, and direct brain and CNS targeting. Due to low bioavailability into the CNS, the BBB has been hindering the evolution of several promising CNS therapeutic candidates. Because the nose and the CNS have different connections, the olfactory route can transmit active pharmaceutical ingredients substances into the brain by crossing the BBB. The ablility to transfer medications into the brain through drug absorption over the olfactory region of the nose is a distinctive feature. The intranasal mode of administration allows the drug to be absorbed by the systemic circulation (Bahadur S, et al., 2020).
Iontophoretic delivery: The use of the iontophoretic technique for CNS medication delivery has sparked widespread attention. Iontophoresis is a methodology that utilizes an external electric field to transport ionised molecules across the BBB. Iontophoretic devices that use the olfactory pathways to deliver drugs to the brain have been developed. They’ve also been developed to enhance macromolecule agent delivery to the brain and CNS. These devices use ions to increase medicament delivery into the brain under controlled parameters, according to programmed modifica tions. The protein and peptide are transported into the brain through an iontophoretic method (Bourganis V, et al., 2018).
Recent advancements
Nano technological advancement was attributed with the most enhanced transport mechanism for therapeutic agent to release into the intended site. The carrier secures and contributes to the improvement of the encapsulated therapeutic ingredient by insulating it from external degradation (Prajapati JI, et al., 2012; Burgess A and Hynynen K, 2013).
According to literature survey, PEGylation (Polyethylene Glycol) is a degree of chemical treatment boosting the biocompatibility of cationic Poly Amido Amine (PAMAM) dendrimers (Joseph E and Saha RN, 2013). The PEGylated PAMAM dendrimers did not affect the integrity of the BBB in an in vitromodel, and they did not trigger cytotoxicity in hypoxia induced by oxygen-glucose deprivation. The PEGylated dendrimers also have the ability to prevent blood clotting, which was an added benefit in stroke treatment (Serlin Y, et al., 2015). The enhanced PAMAM formulation was found in the brain 24 hours after administration in a chronic brain ischemia animal model. The PEGylated PAMAM dendrimers improve blood circulation’s half-life and could be utilized in mucoadhesive drug delivery systems.
Recent studies focus on the development of injectable hydrogels as they can be directly injected into the stroke cavity. As injected hydrogel can easily bypass BBB and promotes local regeneration (Li Y, et al., 2017). They can be used parallel used as transplantation carriers. Recently work is undergone in the manufacturing of contact lenses, tissue engineering scaffolds, wound healing, etc.
Discussion and Conclusion
The preceding demonstrates that nanoparticulate systems have significant promise for converting poorly soluble, poorly absorbed, and labile physiologically active substances into promising drug delivery methods. Because of the hydrophilic shell that hinders identification by the reticular-endothelial system, the core of this system may include a variety of medicines, enzymes, and genes and has lengthy circulation duration. Greater understandings of the many processes of biological interactions, as well as particle engineering, are still necessary to enhance this drug delivery method. To develop the notion of nanoparticle technology into a viable practical application as the next generation of medication delivery system, further progress is required. Due to anatomic and pathophysiological brain barriers, brain-targeted drug delivery is challenging. Current developments in nanotechnology have provided a solution in the form of nanopharmaceuticals, which are drug-containing nanocarriers that can bypass CNS barriers and target brain tissue in a variety of illnesses. The mechanism of delivery of nanopharmaceuticals into the body is a significant factor that influences drug concentration in the brain and drug therapeutic impact. The methods through which nanopharmaceuticals can be administered to the brain. Because of their unique physicochemical features and surface modification, nanopharmaceuticals are able to circumvent route-related restrictions in drug delivery to the brain. Passage of drug molecules through the BBB and BCSF, drug delivery to the brain for the treatment of various ailments is extremely complicated and difficult. To solve these issues and obstacles, techniques such as invasive, non-invasive, intranasal medication administration, ocular drug delivery, and focused ultrasound technique are developed to target the brain. They find things simpler for the therapeutic molecule to cross the BBB and CSF, improving treatment efficacy. The existing techniques, approaches for brain targeted delivery of drug, and significant developments in the last few years were all discussed in this review study.
References
- Taghipour YD, Bahramsoltani R, Marques AM, Naseri R, Rahimi R, Haratipour P, et al. A systematic review of nano formulation of natural products for the treatment of inflammatory bowel disease: Drug delivery and pharmacological targets. DARU. 2018; 26(2): 229-239.
[Crossref] [Google Scholar] [Pubmed]
- Patra JK, Das G, Fraceto LF, Campos EV, Rodriguez-Torres MD, Acosta-Torres LS, et al. Nano based drug delivery systems: Recent developments and future prospects. J Nanobiotechnology. 2018; 16(1):1-33.
[Crossref] [Google Scholar] [Pubmed]
- Lin LN, Liu Q, Song L, Liu FF, Sha JX. Recent advances in nanotechnology based drug delivery to the brain. Cytotechnology. 2010; 62(5): 377-380.
[Crossref] [Google Scholar] [Pubmed]
- Rangasamy M. Nano technology: A review. J Appl Pharm Sci. 2011; 1(2): 8-16.
- González-Nieto D, Fernández-Serra R, Pérez-Rigueiro J, Panetsos F, Martinez-Murillo R, Guinea GV. Biomaterials to neuroprotect the stroke brain: A large opportunity for narrow time windows. Cells. 2020; 9(5): 1074.
[Crossref] [Google Scholar] [Pubmed]
- Tian X, Fan T, Zhao W, Abbas G, Han B, Zhang K, et al. Recent advances in the development of nanomedicines for the treatment of ischemic stroke. Bioact Mater. 2021; 6(9): 2854-2869.
[Crossref] [Google Scholar] [Pubmed]
- Johanson CE, Duncan JA, Stopa EG, Baird A. Enhanced prospects for drug delivery and brain targeting by the choroid plexus-CSF route. Pharm Res. 2005; 22(7): 1011-1037.
[Crossref] [Google Scholar] [Pubmed]
- Johanson CE. Permeability and vascularity of the developing brain: Cerebellum vs. cerebral cortex. Brain Res. 1980; 190(1): 3-16.
[Crossref] [Google Scholar] [Pubmed]
- Biddlestone-Thorpe L, Marchi N, Guo K, Ghosh C, Janigro D, Valerie K, et al. Nanomaterial-mediated CNS delivery of diagnostic and therapeutic agents. Adv Drug Deliv Rev. 2012; 64(7): 605-613.
[Crossref] [Google Scholar] [Pubmed]
- Laquintana V, Trapani A, Denora N, Wang F, Gallo JM, Trapani G. New strategies to deliver anticancer drugs to brain tumors. Expert Opin Drug Deliv. 2009; 6(10): 1017-1032.
[Crossref] [Google Scholar] [Pubmed]
- Ohgaki H, Kleihues P. Genetic pathways to primary and secondary glioblastoma. Am J Patholy. 2007; 170(5): 1445-1453.
[Crossref] [Google Scholar] [Pubmed]
- Burgess A, Hynynen K. Noninvasive and targeted drug delivery to the brain using focused ultrasound. ACS Chem Neurosci. 2013; 4(4): 519-526.
[Crossref] [Google Scholar] [Pubmed]
- Komarova Y, Malik AB. Regulation of endothelial permeability via paracellular and transcellular transport pathways. Annu Rev Physiol. 2010; 72: 463-493.
[Crossref] [Google Scholar] [Pubmed]
- Ghersi‐Egea JF, Strazielle N. Brain drug delivery, drug metabolism, and multidrug resistance at the choroid plexus. Microsc Res Tech. 2001; 52(1): 83-88.
[Crossref] [Google Scholar] [Pubmed]
- Rautio J, Kumpulainen H, Heimbach T, Oliyai R, Oh D, Järvinen T, et al. Prodrugs: design and clinical applications. Nat Rev Drug Discov. 2008; 7(3): 255-270.
[Crossref] [Google Scholar] [Pubmed]
- Rodriguez A, Tatter SB, Debinski W. Neurosurgical techniques for disruption of the blood-brain barrier for glioblastoma treatment. Pharmaceutics. 2015; 7(3): 175-187.
[Crossref] [Google Scholar] [Pubmed]
- Wallez Y, Huber P. Endothelial adherens and tight junctions in vascular homeostasis, inflammation and angiogenesis. Biochim Biophys Acta. 2008; 1778(3): 794-809.
[Crossref] [Google Scholar] [Pubmed]
- Rao KS, Reddy MK, Horning JL, Labhasetwar V. TAT-conjugated nanoparticles for the CNS delivery of anti-HIV drugs. Biomaterials. 2008; 29(33): 4429-4438.
[Crossref] [Google Scholar] [Pubmed]
- Taddei A, Giampietro C, Conti A, Orsenigo F, Breviario F, Pirazzoli V, et al. Endothelial adherens junctions control tight junctions by VE-cadherin-mediated upregulation of claudin-5. Nat Cell Biol. 2008; 10(8): 923-934.
[Crossref] [Google Scholar] [Pubmed]
- Pober JS, Sessa WC. Evolving functions of endothelial cells in inflammation. Nat Rev Immunol. 2007; 7(10): 803-815.
[Crossref] [Google Scholar] [Pubmed]
- Erdő F, Bors LA, Farkas D, Bajza Á, Gizurarson S. Evaluation of intranasal delivery route of drug administration for brain targeting. Brain Res Bull. 2018; 143: 155-170.
[Crossref] [Google Scholar] [Pubmed]
- Bahadur S, Naurange T, Baghel P, Sahu M, Yadu K. Targeting the brain: Various approaches and science involved. Pharm Sci. 2020; 5 (27): 4-16.
- Teleanu DM, Chircov C, Grumezescu AM, Volceanov A, Teleanu RI. Blood-brain delivery methods using nanotechnology. Pharmaceutics. 2018; 10(4): 269.
[Crossref] [Google Scholar] [Pubmed]
- Mehdipour AR, Hamidi M. Brain drug targeting: A computational approach for overcoming blood-brain barrier. Drug Discov today. 2009; 14(21-22): 1030-1036.
[Crossref] [Google Scholar] [Pubmed]
- Radocha J, van de Donk NW, Weisel K. Monoclonal antibodies and antibody drug conjugates in multiple myeloma. Cancers. 2021; 13(7): 1571.
[Crossref] [Google Scholar] [Pubmed]
- Slavc I, Cohen-Pfeffer JL, Gururangan S, Krauser J, Lim DA, Maldaun M, et al. Best practices for the use of intracerebroventricular drug delivery devices. Mol Genet Metab. 2018; 124(3): 184-188.
[Crossref] [Google Scholar] [Pubmed]
- Ningaraj NS, Rao M, Hashizume K, Asotra K, Black KL. Regulation of blood-brain tumor barrier permeability by calcium-activated potassium channels. J Pharmacol Exp Ther. 2002; 301(3): 838-851.
[Crossref] [Google Scholar] [Pubmed]
- Avhad PS, Patil PB, Jain NP, Laware SG. A Review on Different Techniques for Brain Targeting. Int J Pharm Chem Anal. 2015; 2(3): 143-147.
- Dixit N, Bali V, Baboota S, Ahuja A, Ali J. Iontophoresis-an approach for controlled drug delivery: A review. Curr Drug Deliv. 2007; 4(1): 1-10.
[Crossref] [Google Scholar] [Pubmed]
- Ghersi-Egea JF, Strazielle N. Choroid plexus transporters for drugs and other xenobiotics. J Drug Target. 2002; 10(4): 353-357.
[Crossref] [Google Scholar] [Pubmed]
- Bourganis V, Kammona O, Alexopoulos A, Kiparissides C. Recent advances in carrier mediated nose-to-brain delivery of pharmaceutics. Eur J Pharm Biopharm. 2018; 128: 337-362.
[Crossref] [Google Scholar] [Pubmed]
- Prajapati JI, Patel HA, Agrawal YK. Targeted drug delivery for central nervous system: A review. Int J Pharm Pharm Sci. 2012; 3: 32-38.
- Burgess A, Hynynen K. Noninvasive and targeted drug delivery to the brain using focused ultrasound. ACS Chem Neurosci. 2013; 4(4): 519-526.
[Crossref] [Google Scholar] [Pubmed]
- Joseph E, Saha RN. Advances in brain targeted drug delivery: Nanoparticulate systems. J PharmaSciTech. 2013; 3(1): 1-8.
- Serlin Y, Shelef I, Knyazer B, Friedman A. Anatomy and physiology of the blood-brain barrier. Semin Cell Dev Biol. 2015; 38: 2-6.
[Crossref] [Google Scholar] [Pubmed]
- Li Y, Chen T, Miao X, Yi X, Wang X, Zhao H, et al. Zebrafish: A promising in vivo model for assessing the delivery of natural products, fluorescence dyes and drugs across the blood-brain barrier. Pharmacol Res. 2017; 125: 246-257.
[Crossref] [Google Scholar] [Pubmed]
Author Info
K Sree Lakshmi*Citation: Lakshmi KS: A Review on Nanoformulations Targeting Brain Drug Delivery
Received: 01-Jul-2022 Accepted: 25-Jul-2022 Published: 01-Aug-2022, DOI: 10.31858/0975-8453.13.8.563-566
Copyright: This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
ARTICLE TOOLS
- Dental Development between Assisted Reproductive Therapy (Art) and Natural Conceived Children: A Comparative Pilot Study Norzaiti Mohd Kenali, Naimah Hasanah Mohd Fathil, Norbasyirah Bohari, Ahmad Faisal Ismail, Roszaman Ramli SRP. 2020; 11(1): 01-06 » doi: 10.5530/srp.2020.1.01
- Psychometric properties of the World Health Organization Quality of life instrument, short form: Validity in the Vietnamese healthcare context Trung Quang Vo*, Bao Tran Thuy Tran, Ngan Thuy Nguyen, Tram ThiHuyen Nguyen, Thuy Phan Chung Tran SRP. 2020; 11(1): 14-22 » doi: 10.5530/srp.2019.1.3
- A Review of Pharmacoeconomics: the key to “Healthcare for All” Hasamnis AA, Patil SS, Shaik Imam, Narendiran K SRP. 2019; 10(1): s40-s42 » doi: 10.5530/srp.2019.1s.21
- Deuterium Depleted Water as an Adjuvant in Treatment of Cancer Anton Syroeshkin, Olga Levitskaya, Elena Uspenskaya, Tatiana Pleteneva, Daria Romaykina, Daria Ermakova SRP. 2019; 10(1): 112-117 » doi: 10.5530/srp.2019.1.19
- Dental Development between Assisted Reproductive Therapy (Art) and Natural Conceived Children: A Comparative Pilot Study Norzaiti Mohd Kenali, Naimah Hasanah Mohd Fathil, Norbasyirah Bohari, Ahmad Faisal Ismail, Roszaman Ramli SRP. 2020; 11(1): 01-06 » doi: 10.5530/srp.2020.1.01
- Manilkara zapota (L.) Royen Fruit Peel: A Phytochemical and Pharmacological Review Karle Pravin P, Dhawale Shashikant C SRP. 2019; 10(1): 11-14 » doi: 0.5530/srp.2019.1.2
- Pharmacognostic and Phytopharmacological Overview on Bombax ceiba Pankaj Haribhau Chaudhary, Mukund Ganeshrao Tawar SRP. 2019; 10(1): 20-25 » doi: 10.5530/srp.2019.1.4
- A Review of Pharmacoeconomics: the key to “Healthcare for All” Hasamnis AA, Patil SS, Shaik Imam, Narendiran K SRP. 2019; 10(1): s40-s42 » doi: 10.5530/srp.2019.1s.21
- A Prospective Review on Phyto-Pharmacological Aspects of Andrographis paniculata Govindraj Akilandeswari, Arumugam Vijaya Anand, Palanisamy Sampathkumar, Puthamohan Vinayaga Moorthi, Basavaraju Preethi SRP. 2019; 10(1): 15-19 » doi: 10.5530/srp.2019.1.3






