Research Article - (2022) Volume 13, Issue 3
Abstract
Introduction: The relationship between trace elements and Oxidative Stress (OS) in Chronic Kidney Disease (CKD) patients is still not completely elucidated. The aim of this work is to determine the serum levels of OS and the trace elements in CKD patients.
Methods: A total of 91 patients were enrolled and then divided into 5 groups at the basis of CKD stages and therapy methods. Healthy volunteers were included as a control group. Serum levels of SOD, MDA, Zn, Cu, Ca, Fe, and Mg were determined, and the correlations among these data’s were analyzed; a portion of patients are under hemodialysis and peritoneal dialysis. Meanwhile, the association between OS and trace elements will be investigated.
Results: The Malondialdehyde (MDA) level in the patients’ sera was significantly higher than the controls’ (P<0.05); however, their sera level of Superoxide Dismutase (SOD) was obviously lower (P<0.05). Meanwhile, the creatinine level in CKD patients’ sera was positively correlated with the MDA level (r=0.534, P<0.01), while negatively with the expression of SOD (r=-0.427, P<0.01). A lower expression of Zn and Ca values could be seen in the cases’ sera (P<0.05). Besides that, it showed a significant negative correlation between serum levels of MDA and Ca (r=-0.282, P<0.01), MDA and Zn (r=-0.358, P<0.01), while a positive correlation between MDA and Cu (r=0.236, P=0.022). Adversely, the expression of SOD in the sera of cases was positively related to the levels of Ca (r=0.273, P<0.01) and Zn (r=0.277, P<0.01).
Conclusion: Abnormalities of OS and Zn, Ca existed in CKD patients; and elevated OS may play a role in the trace elements imbalance. Dialysis not only hemodialysis but also peritoneal dialysis seems to have no obvious effect on plasma trace elements. Correct the disorders need to find new ways.
Keywords
Chronic kidney disease, Oxidative stress, Trace element, Dialysis, Hemodialysis
Introduction
Chronic Kidney Disease (CKD) is characterized by progressive loss of kidney function, and has turned into a major global health problem. The incidence and mortality of CKD have increased over the past few decades (Lozano R, et al., 2012). A Chinese survey in 2012 found that the overall prevalence of CKD in adults was approximately 10.8%, placing a huge burden on health care resources (Zhang L, et al., 2012).
Oxidative Stress (OS) refers to an imbalance between formation of Reactive Oxygen Species (ROS) and antioxidative defense mechanisms. In CKD patients, Oxidative Stress is an important risk factor for the development and progression of several complications. There is good evidence indicating that uremia in general is associated with enhanced Oxidative Stress, and treatment of uremic patients with Hemodialysis group (HD) has been suggested to particularly contribute to Oxidative Stress and reduced antioxidant levels (Galle J, 2001; Oberg BP, et al., 2004).
Trace elements, which are measured in microgram per gram or less, occur in minute amounts and are indispensable for our body. In some instances, they are the key components of many enzymatic pathways (Yavuz O, et al., 2004), thereby play important physiological and biochemical functions. Many studies confirmed that patients with CKD are often at risks of developing severe trace elements imbalances (Locatelli F, et al., 2003; Sandstead HH, 1980).
Dialysis, including Hemodialysis and Peritoneal Dialysis, is the most common and significant method of treatment for End-Stage Renal Disease (ESRD). Researches in regard to the changes of trace elements in the blood levels have been reported in Chronic Renal Failure (CRF) patients, especially in those treated by Hemodialysis (Vanholder R, et al., 2002). The effect of trace elements on metabolism may be exacerbated in ESRD patients on dialysis because these patients are incapable of excreting excess elements properly (Covic A and Gusbeth-Tatomir P, 2009). However, the incidence of abnormal trace element status in different stages of CKD patients, including CKD3, CKD4, CKD5, especially in dialysis patients, has not been comprehensively studied.
In this study, we investigate the plasma trace elements levels and OS parameters in CKD patients, determining plasma levels of Zn, Cu, Ca, Fe and Mg. We also evaluated plasma Malondialdehyde (MDA) and Superoxide Dismutase (SOD), which are the key markers of OS. The relation between OS and those trace elements are investigated as well.
Materials and Methods
Patients
All cases were recruited from the Department of Nephrology and Blood Purification Center in the Second Affiliated Hospital of Xi’an Jiaotong University. The primary disease for all patients was chronic glomerulonephritis, and the inclusion criteria were as follows: Stable blood pressure, absence of complicated infection and heart failure within the last 3 months, not pregnant or breast feeding, absence of severe cardiovascular and mental disorders. The patients were not taking any immunosuppressive agents.
A total of 91 patients were enrolled and divided into 3 groups based on the CKD stages calculated from Glomerular Filtration Rate (GFR). Using the Modification of Diet in Renal Disease (MDRD) equation to calculate the estimated GFR. Patients are grouped into CKD3, CKD 4 and CKD5 which were further divided into 3 subgroups: Non-dialysis uremic group (NHD), Hemodialysis group (HD) and Peritoneal Dialysis group (PD). The patients in the HD group had undergone regular hemodialysis treatment (4-hour treatment given 3 times a week) for more than 3 months using a dialysis solution (sugar-free, Ca2+ 1.5 mmol/L, K+ 2.0 mmol/L). The dialysis membrane was polysulfone with an area of 1.4-1.8 m2, and the blood flow was set to 220-250 mL/min, dialysate flow 500 mL/min. The PD patients had undergone regular treatment for more than 3 months using low-calcium Peritoneal Dialysis solution, which were the standard Tenckhoff catheter, and the daily volume of replacement fluid was 8000 mL. Twenty healthy volunteers were recruited to the cont rol group.
This study was approved by the Ethics committee in the Second Affiliated Hospital of Xi’an Jiaotong University. Written informed consent was obtained from all patients and healthy volunteers participating in the study.
Sample preparation
Fasting hemospasia was performed in all participants. Blood samples in the HD group were collected prior to Hemodialysis on the day of treatment. Routine blood test that included a complete blood count and routine clinical chemistry tests were performed. 10 mL of peripheral blood was centrifuged at 3000 rpm for 5 min, and the supernatant was collected and stored at -80°C.
Enzyme-Linked Immunosorbent Assay (ELISA)
All serum samples were stored at -80ºC and processed all at once. Serum levels of Cu, Ca, Fe, Zn, Mg (Xi’an microelement inspection institute), SOD and MDA (Westang, Shanghai, China) were determined by Enzyme-Linked Immunosorbent Assay (ELISA) using specific detection reagents following the manufacturer’s instruction. Each assay was performed in triplicates and the experiments were repeated once.
Statistical analysis
Data were analyzed using SPSS 16.0 Statistical Analysis software (SPSS Inc., Chicago, IL, USA). Measurement data are expressed as mean ± Standard Deviation (SD). Comparison of means among groups was made by one-way Analysis of Variance (ANOVA), and comparison between two groups was made by Fisher's Least Significant Difference (LSD) test. Spearman’s rank-order correlation was used for bivariate correlation analysis. A P value less than 0.05 (P<0.05) was considered statistically significant, and P<0.01 indicated a more significant difference.
Results
Patients characteristics
Clinical characteristics of CKD patients: A total of 91 patients were classified based on the following CKD stages: CKD 3 (12 males and 10 females, mean age: 58.9 ± 19.0 years), CKD 4 (10 males and 9 females, mean age: 58.3 ± 13.1 years), and CKD 5 which were further divided into NHD group (8 males and 6 females, mean age: 54 ± 20.1 years), HD group (9 males and 9 females, mean age: 48.3 ± 17.5 years) and PD group (9 males and 9 females, mean age: 46.6 ± 15.0 years). Healthy volunteers were recruited to the control group (12 males and 8 females, mean age: 48.9 ± 11.4 years).
Routine blood and clinical chemistry tests were performed. Complete blood count was measured. Results were analyzed for each group and comparison was made between groups (Table 1). Significant differences between the controls and different stage of CKD patients were observed in parameters (Table 1). Also, there were significant differences of hemoglobin, serum creatinine, urea nitrogen, cystatin-C, parathyroid hormone among CKD groups (Table 2).
| Item | Control(n=20) | CKD3(n=22) | CKD4(n=19) | NHD(n=14) | CKD5(n=50) | PD(n=18) |
|---|---|---|---|---|---|---|
| HD(n=18) | ||||||
| Age | 48.9 ± 11.4 | 58.9 ± 19.0 | 58.3 ± 13.1 | 54.0 ± 20.1 | 48.3 ± 17.5 | 46.6 ± 15.0 |
| Gender (male/female) | 12/8 | 12/10 | 10/9 | 8/6 | 9/9 | 10/8 |
| WBC (1012/L) | 6.17 ± 1.67 | 7.06 ± 3.21 | 5.57 ± 1.57# | 5.65 ± 1.63 | 5.64 ± 1.28 | 6.07 ± 2.10 |
| RBC (1012/L) | 4.62 ± 0.57 | 3.46 ± 0.82* | 2.81 ± 0.65*# | 2.55 ± 0.68*# | 3.30 ± 0.72*& | 2.89 ± 0.69*# |
| PLT (109/L) | 235 ± 51.7 | 161 ± 58.5* | 167 ± 61.4* | 145 ± 60.7* | 157 ± 46.3* | 164 ± 94.0* |
| Hb (g/L) | 135 ± 15.5 | 104 ± 26.0* | 82.8 ± 18.1* | 77.5 ± 12.7*# | 101 ± 21.3* ※& | 85.3 ± 20.0*# |
| FPG (mmol/L) | 5.10 ± 0.74 | 5.10 ± 0.98 | 4.75 ± 0.52 | 4.86 ± 2.09 | 4.28 ± 0.76$ | 5.47 ± 2.56 |
| Scr (μmol/L) | 69.6 ± 11.6 | 267.2 ± 112.3* | 556.6 ± 80.3*# | 983.4 ± 372.7*#※ | 897.3 ± 242.2*# ※ | 851.6 ± 218*# ※ |
| BUN (mmol/L) | 4.30 ± 0.93 | 14.5 ± 7.01* | 20.0 ± 5.94*# | 25.8 ± 9.50*# ※ | 26.1 ± 7.64*# ※ | 20.7 ± 8.12*# |
| Cystre-C (mg/L) | 0.77 ± 0.16 | 3.42 ± 1.90* | 5.31 ± 1.86*# | 5.84 ± 1.94*# | 8.22 ± 4.10*# ※&$ | 6.06 ± 1.09*# |
| UA (μmol/L) | 276.0 ± 89.4 | 400.7 ± 136.4* | 370.7 ± 108.1* | 412.2 ± 158.4* | 400.2 ± 83.8* | 434.2 ± 119.8* |
| PTH (pg/mL) | 38.0 ± 14.8 | 129.3 ± 131.7* | 203.8 ± 87.1* | 274.7 ± 129.2*# | 397.3 ± 349.5*# ※ | 319.5 ± 136.9*# ※ |
| ALT (IUI/L) | 25.8 ± 10.8 | 18.3 ± 15.3 | 17.9 ± 12.8 | 20.7 ± 9.6 | 12.8 ± 6.1 | 13.4 ± 7.1 |
| AST (IUI/L) | 19.4 ± 3.8 | 23.7 ± 17.8 | 19.8 ± 8.9 | 23.5 ± 7.2 | 14.1 ± 5.1 | 15.8 ± 6.2 |
| TP (g/L) | 71.6 ± 3.26 | 64.4 ± 9.68* | 66.9 ± 9.0 | 62.1 ± 7.82* | 65.3 ± 4.64* | 54.5 ± 5.17*# ※&§ |
| ALB(g/L) | 45.3 ± 2.73 | 38.4 ± 6.43* | 38.9 ± 6.69* | 37.7 ± 5.93* | 39.5 ± 4.39* | 29.9 ± 4.76*# ※&§ |
| Ca (mmol/L) | 2.01 ± 1.09 | 2.03 ± 0.34 | 1.95 ± 0.29 | 1.87 ± 0.22 | 1.92 ± 0.29 | 1.67 ± 0.23*# ※§ |
| K (mmol/L) | 4.32 ± 0.44 | 4.55 ± 0.79 | 4.55 ± 0.96 | 5.07 ± 0.65* | 5.28 ± 0.76*# ※ | 3.90 ± 1.16# ※&§ |
| CRP (mg/L) | 6.8 ± 0.66 | 12.6 ± 0.78* | 15.7 ± 0.58* | 18.2 ± 0.46*# | 19.4 ± 0.57*# | 15.7 ± 0.61* |
| TC (mmol/L) | 4.45 ± 0.97 | 4.50 ± 1.13 | 4.40 ± 1.34 | 3.94 ± 0.48 | 3.72 ± 0.67*# | 4.14 ± 0.95 |
| TG (mmol/L) | 1.85 ± 1.39 | 1.99 ± 1.24 | 1.34 ± 0.94 | 1.28 ± 0.31# | 1.13 ± 0.44# | 1.12 ± 0.59# |
Abbreviation: Values are expressed as mean ± SD (Standard Deviation). RF: Renal Failure; NHD: Not Hemodialysis; HD: Hemodialysis; PD: Peritoneal Dialysis; Scr: Serum creatinine; BUN: Urea Nitrogen; WBC: Leukocyte; RBC: Erythrocyte; PLT: Blood; Hb: Hemoglobin; TP: Total Protein; UA: Uric Acid; ALB: Albumin; TC: Total Cholesterol; TG: Triglyceride; PTH: Parathyroid Hormone; FPG: Fasting Plasma Glucose; ALT: Glutamic-pyruvic transaminase; AST: Glutamic oxalacetic transaminase; CRP: C-Reaction Protein; CKD: Chronic Kidney Disease. *p<0.05 vs. control; #p<0.05 vs. CKD3; ※p<0.05 vs. CKD4; &p<0.05 vs. NHD; §p<0.05 vs. HD; $p<0.05 vs. PD. All the differences were calculated by the Least Significant Difference (LSD) test.
Table 1: Clinical characteristics of all CKD patients
| Item | Control (n=20) | CKD3 (n=22) | CKD4 (n=19) | CKD5(n=50) | ||
|---|---|---|---|---|---|---|
| NHD(n=14) | HD (n=18) | PD (n=18) | ||||
| SOD | 473.62 ± 231.08 | 315.79 ± 153.18 | 248.32 ± 173.96* | 168.12 ± 98.89*# | 237.39 ± 117.17* | 204.53 ± 124.47* |
| MDA | 201.15 ± 52.43 | 349.26 ± 66.17* | 426.28 ± 179.28* | 488.73 ± 158.34*# | 434.49 ± 109.12*# | 411.86 ± 113.5* |
Note: *p<0.05 vs. control; #p<0.05 vs. CKD3; ※p<0.05 vs. CKD4; & p<0.05 vs. NHD; §p<0.05 vs. HD; $p<0.05 vs. PD. All the differences were calculated by the LSD test. SOD: Superoxide Dismutase; MDA: Malondialdehyde; PD: Peritoneal Dialysis; HD: Hemodialysis; NHD: Not Hemodialysis; CKD: Chronic Kidney Disease. LSD: Least Significant Difference; OS: Oxidative Stress.
Table 2: Serum concentrations of OS markers in CKD patients and normal control subjects
There were no significant differences in age and gender distributions between the controls and patients in different CKD stages (Table 3).
| Item | Control (n=20) | CKD3 (n=22) | CKD4 (n=19) | CKD5(n=50) | ||
|---|---|---|---|---|---|---|
| NHD(n=14) | HD (n=18) | PD (n=18) | ||||
| Zn | 0.925 ± 0.135 | 0.746 ± 0.290* | 0.781 ± 0.252 | 0.815 ± 0.247 | 0.655 ± 0.278* | 0.607 ± 0.259* |
| Fe | 1.280 ± 0.602 | 1.383 ± 0.490 | 1.377 ± 0.546 | 1.334 ± 0.662 | 1.345 ± 0.430 | 1.423 ± 0.482 |
| Ca | 81.06 ± 4.504 | 74.97 ± 13.30 | 73.51 ± 7.470* | 68.66 ± 11.02* | 72.15 ± 9.54* | 65.27 ± 8.420*#※ |
| Mg | 19.48 ± 2.403 | 22.50 ± 4.037 | 21.73 ± 4.717 | 21.27 ± 4.666 | 22.82 ± 4.589* | 21.91 ± 4.25 |
| Cu | 1.222 ± 0.368 | 1.336 ± 0.487 | 1.475 ± 0.411 | 1.398 ± 0.361 | 1.280 ± 0.454 | 1.300 ± 0.416 |
Note: *p<0.05 vs. control; #p<0.05 vs. CKD3; ※p<0.05 vs. CKD4; &p<0.05 vs. NHD; §p<0.05 vs. HD; $p<0.05 vs. PD. All the differences were calculated by the LSD test. PD: Peritoneal Dialysis; HD: Hemodialysis; NHD: Not Hemodialysis; CKD: Chronic Kidney Disease; LSD: Least Significant Difference.
Table 3: Micro-trace elements in Chronic Renal Failure (CRF) patients and normal control subjects (ug/ml)
Expression profiles of serum cytokines: We evaluated the correlation serum levels of the OS markers with creatinine levels. Serum levels of SOD (Figure 1A) and MDA (Figure 1B) were respectively significantly lower and higher in CKD patients than in controls (P<0.05), and positively correlated with advancing CKD stage (P<0.05). Serum levels of SOD were altered by dialysis treatment compared with no-dialysis patients. In contrast, serum levels of MDA were not altered by dialysis treatment.
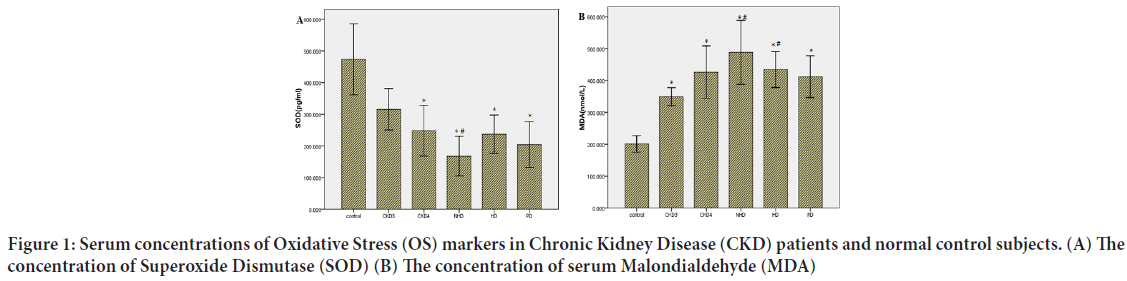
Figure 1: Serum concentrations of Oxidative Stress (OS) markers in Chronic Kidney Disease (CKD) patients and normal control subjects. (A) The concentration of Superoxide Dismutase (SOD) (B) The concentration of serum Malondialdehyde (MDA)
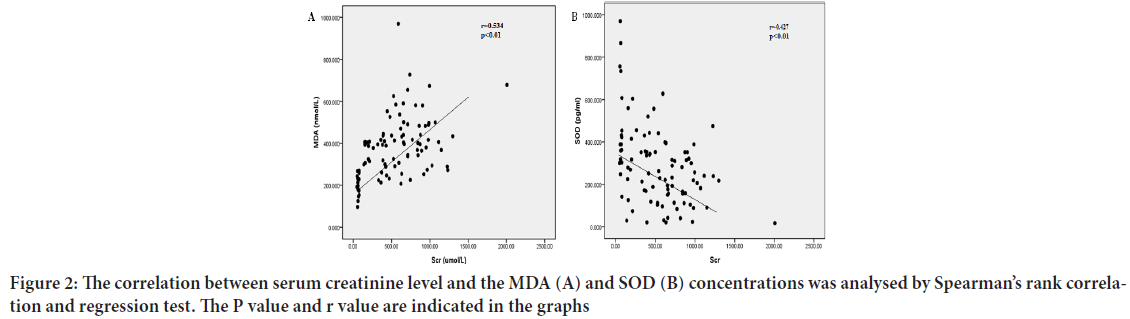
Moreover, serum creatinine levels in serum CKD patients correlated positively with serum MDA levels (r=0.534, P<0.01), and negatively with serum SOD levels (r=-0.427, P<0.01) (Figures 2A and 2B), reflecting an association between the extent of OS and the severity of renal damage.
Figure 2: The correlation between serum creatinine level and the MDA (A) and SOD (B) concentrations was analysed by Spearman’s rank correlation and regression test. The P value and r value are indicated in the graphs
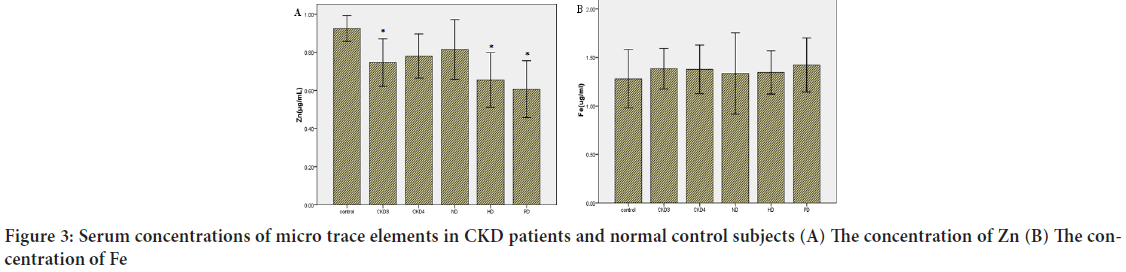
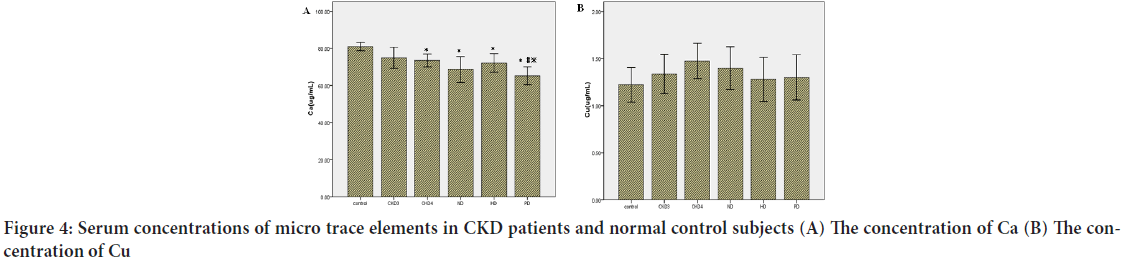
Micro trace elements: Serum levels of Zn (Figure 3A), Fe (Figure 3B), Ca (Figure 4A), Cu (Figure 4B) and Mg were analyzed by ELISA. The serum levels of Zn and Ca were significantly lower in CKD patients than controls (P<0.05). Serum levels of Zn were altered by dialysis treatment compared with no-dialysis. In contrast, serum levels of Ca were not altered by dialysis treatment. There were no significant differences in Fe, Cu, and Mg between CKD patients and controls.
Figure 3: Serum concentrations of micro trace elements in CKD patients and normal control subjects (A) The concentration of Zn (B) The concentration of Fe
Figure 4: Serum concentrations of micro trace elements in CKD patients and normal control subjects (A) The concentration of Ca (B) The concentration of Cu
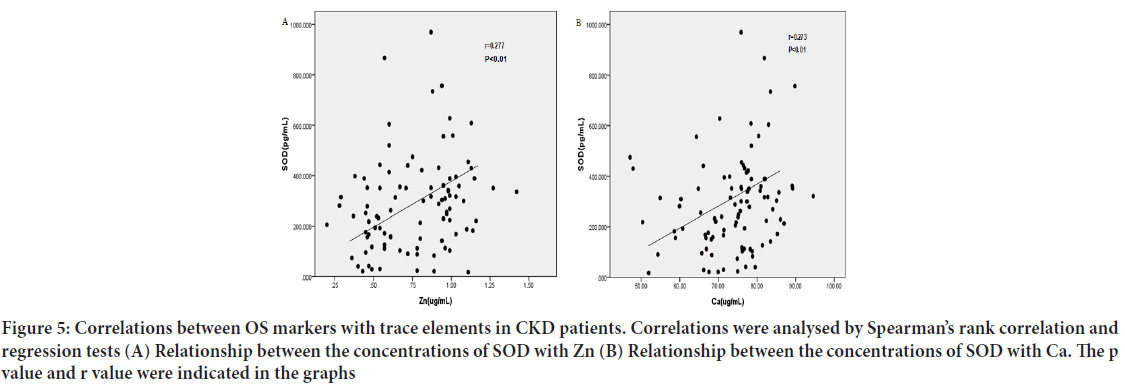
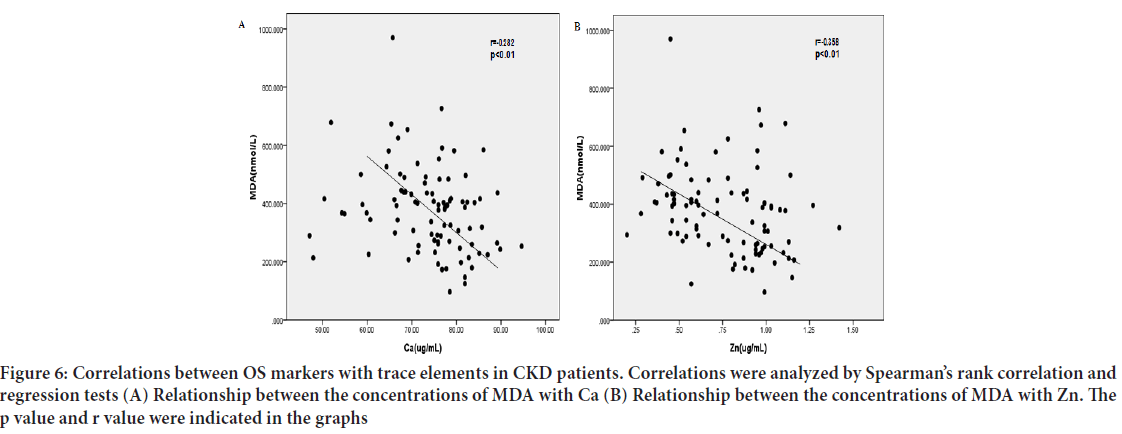
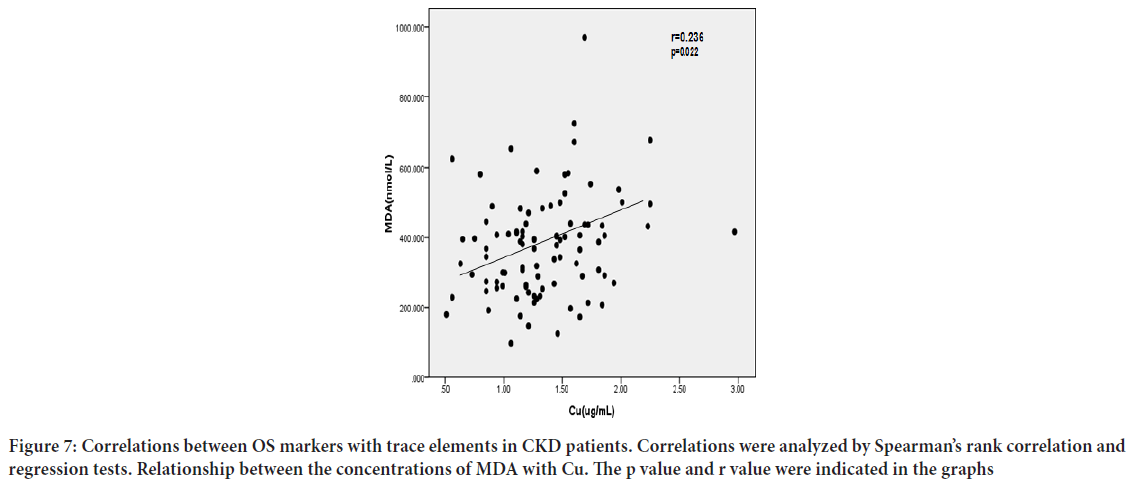
Correlation between SOD, MDA with Micro trace elements: There was a significant and positive correlation between serum levels of SOD and Zn (Figure 5A) (r=0.277, P<0.01), Ca (Figure 5B) r=0.273, P<0.01. Adversely, there was a significant and negative correlation between MDA and Ca (Figure 6A) r=-0.282, P<0.01, Zn (Figure 6B) (r=-0.358, P<0.01), as well as a positive correlation between MDA and serum Cu, which did not quite reach statistical significance (Figure 7) (r=0.236, P=0.022). Other trace elements have no obvious correlation with SOD and MDA.
Figure 5: Correlations between OS markers with trace elements in CKD patients. Correlations were analysed by Spearman’s rank correlation and regression tests (A) Relationship between the concentrations of SOD with Zn (B) Relationship between the concentrations of SOD with Ca. The p value and r value were indicated in the graphs
Figure 6: Correlations between OS markers with trace elements in CKD patients. Correlations were analyzed by Spearman’s rank correlation and regression tests (A) Relationship between the concentrations of MDA with Ca (B) Relationship between the concentrations of MDA with Zn. The p value and r value were indicated in the graphs
Figure 7: Correlations between OS markers with trace elements in CKD patients. Correlations were analyzed by Spearman’s rank correlation and regression tests. Relationship between the concentrations of MDA with Cu. The p value and r value were indicated in the graphs
Discussion
With recent advances studies of pathophysiology, inflammation and dysregulated immune responses have emerged as important factors contributing to the gradual loss of renal function in CKD patients (Tonelli M, et al., 2009). Moreover, the risk of heart diseases and cancers increases markedly in CKD patients (Ng AH, et al., 2004; Miyamoto T, et al., 2011). Also, the CKD patients are at the risk of developing severe trace elements imbalances (Foley RN, 2008; Collins AJ, et al., 2015). Trace elements are indispensable and often combined with proteins, coenzyme, hormones and vitamins. In ESRD patients, many different factors affect the serum concentrations of trace elements, such as increased oral intake, failure of renal excretion, loss of appetite and metabolic alterations and so on. During dialysis processes some trace elements can accumulate because of dialysis fluid impurities, and others may move from blood to the dialysate, leading to deficiency (Esfahani ST, et al., 2006).
In our study, plasma Fe, Cu and Mg trace element levels of CKD patients, were compared with control group and compared with each other. We found that plasma Fe have no statistically differences between patients and the control group. This may be attributed to the fact that we often supplement iron in CKD patients because most of them have anemia, especially in CKD5. Again there were no difference in plasma Cu and Mg, we analyze that the control and patients had the similar regional changes and dietary habits which soil and water contamination. Adversely, serum Zn and Ca values were significantly lower in our CKD patients than the controls, and the tendency became more obvious as the disease progressed, especially in CKD5 patients. Zn is an essential component of various proteins and biomembrane (Ranaldi G, et al., 2002). Recent studies suggest that, both of the increased urinary zinc excretion and decreased intestinal zinc absorption may be the primary cause of hypozincemia in patients. Also dietary restrictions and anorexia lead to an insufficient micronutrient intake. Shrimpton R, et al., 2005 argue zinc deficiency is a leading cause of diseases in developing countries and is associated with immune deficiencies by impaired cell proliferation, abnormal T-cell function, and cytokine expression, all of which might contribute to the high risk of infection observed in uraemia patients. Meanwhile we found Zn amounts in HD and PD lower than other CKD patients. We think it is because of these patients are more seriously and renal replacement therapies consume more zinc. Ca is one of the most abundant mineral elements in human body, and the normal function of tissues and organs regulating various systems of human body depends on its existence. The person can become sensitive, emotional instability and attention deficit if they lack of Ca. In CKD patients the kidney damage leading to the body of phosphorus excretion disorder on the other hand the diet excessive intake of phosphorus, so combined with Ca induced deposition, it caused the loss of calcium and insufficient production of 1.25-(OH) 2VitD3 which reduce calcium absorption. We found that the decrease in blood calcium is more significant in PD patients which were more likely to use low calcium dialysis fluid. These consequences consistent with several other investigators (Esfahani ST, et al., 2016). Miura Y, et al., 1999 compared serum Mg and Cu levels between HD patients and control groups and found no statistically significant differences. The results of Miura study also found Zn is lower in HD patients. But Krachler M, et al., 2000 found elemental composition changes in plasma and observed a decrease in Mg levels over a 6-month period in uremia (Wirnsberger GH, 2000). Regulating trace element levels is essential to prevent some complications in HD patients. Differing results have been reported with respect to plasma levels of trace elements in HD patients. Tonelli M, et al. found low plasma Zn and high plasma Cu levels in HD patients compared with controls (D'haese PC and de Broe ME, 1996). The distribution of trace elements can be affected by insufficient dietary intake, anorexia, degrees of gastrointestinal absorption, and drug treatment, it lead to some clinical abnormalities in uremic patients (Bhatnagar S and Natchu UC, 2004). The uremic state, medication, the dialysis process, dietary intake, and the quality of water used for dialysis may contribute to such clinical abnormalities of trace elements (Tzanno-Martins C, et al., 1996). Although recent improvements of technology in the dialysis can prevent some serious complications by changes in trace elements, problems still remained (Sanaka T and Koremoto M, 2011). In our study, although dialysis treatment improved the survival of CKD patients, it had a limited effect on trace elements. Dialysis augments reduced renal function by removing metabolic waste products and maintaining the electrolyte balance and acid-base equilibrium. However, due to the possible risks present in HD, such as blood loss during treatment, dietary restrictions, anorexia, aluminum intoxication (Gokal R, et al., 1999) and dialyzer biocompatibility (Vaziri ND, et al., 1998), there is no effect in trace elements balance compared with no-dialysis patients. Although PD has advantages over HD in these respects (Himmelfarb J, et al., 2002), problems such as increased protein loss, excessive glucose intake and triglyceride abnormalities make it ineffective in restoring immune balance. Further investigation is required to identify new therapeutic agents or strategies to improve the patient’s microenvironment.
OS refers to an imbalance in the body oxidation and antioxidation, results in neutrophils inflammatory infiltrates, proteases secretion to increase, and produces a large number of oxide intermediate. Several studies have confirmed that the occurrence of complications in CKD such as accompany hypertension (Vaziri ND, et al., 1998), disease of heart and nervous system lesions, OS is one of the important risk factors affecting prognosis of CKD (Deng G, et al., 2001). SOD and MDA can be used to evaluate the body's resistance to oxidation and peroxidation level. In our study the serum levels of MDA were significantly higher and SOD significantly low er in CKD patients than in controls. Meanwhile we found that serum levels of MDA and SOD were be closely related with serum creatinine levels. The results reiterate that the severity of CKD correlates with the degree of OS. However, as the renal function deteriorates, OS response might impair renal function and the extent of renal damage could conceivably increase during progression of CKD. So we think that the OS reaction which caused by metabolic disorder and apoptosis may be involved in the development process of renal fibrosis. But serum levels of SOD were altered by dialysis treatment compared with no-dialysis, so we think replacement can improve the imbalance in the body oxidation and antioxidation system of CKD patients.
We also compared trace element levels and oxidant-antioxidant parameters and found that in patient groups, there was a significant positive correlation between serum levels of SOD with Zn (Figure 5A) (r=0.277, P<0.01), Ca (Figure 5B) (r=0.273, P<0.01). Adversely, there was a significant negative correlation between serum levels of MDA with Ca (Figure 6A) (r=-0.282, P<0.01), Zn (Figure 6B) (r=-0.358, P<0.01) as well as a positive correlation between MDA and serum Cu, but the relationship did not quite reach statistical significance. According to the above description, OS is one of the important risk factors affecting prognosis of CKD, so we have reasons to regard that Zn and Cu are closely influenced by OS. Cu is a transition element and a cause of radical ROS formation through Fenton reaction. These ROS radicals cause lipid peroxidation, an end product of MDA. Consistent with our study, Rükgauer M, et al., 2001 analyzed trace elements and MDA concentrations in healthy persons and compared Cu levels and MDA concentrations, and found no correlations between them. Other trace elements (Mg and Fe) have no obvious correlation with SOD and MDA.
Conclusion
According to our results, elevate OS may play a role in the trace elements imbalance in CKD. Dialysis itself seems to have no obvious effect on plasma trace elements concentrations compared with No-dialysis CKD5 patients. Therefore, decreasing sources of Oxidative Stress and at the same time increasing oxidative capacity can be beneficial to these patients. These findings show that whereas prolonged replacement treatment leads to improved Oxidative Stress damage, trace element concentrations are not affected. Since both deficiency and excess of trace elements are potentially harmful yet amenable to therapy, the hypothesis that trace element status influences the risk of adverse clinical outcomes is worthy of investigation. And it is critical if micronutrient interventions are to be not only effective, but also targeted to those with the greatest need.
Declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Human Research Committee for Research Involving Human subjects in the Second Affiliated Hospital of Xi’an Jiaotong University and the ethical standards of the institutional and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
The study was approved by the local ethical committee and all patients gave informed consent.
Conflict of interests
The authors declare no financial or commercial conflict of interests.
Acknowledgements
This work was supported by the Science and Technology Program of Xi'an [No.2019114613YX001SF037(2)]. We thank the Department of Nephrology in the Second Affiliated Hospital of Xi'an jiantong Unive rsity for providing the peripheral blood samples. We express our grat itude to all the study participants.
References
- Lozano R, Naghavi M, Foreman K, Lim S, Shibuya K, Aboyans V, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the global burden of disease study 2010. Lancet. 2012; 380(9859): 2095-2128.
[CrossRef] [Google Scholar] [Pubmed]
- Zhang L, Wang F, Wang L, Wang W, Liu B, Liu J, et al. Prevalence of chronic kidney disease in China: A cross-sectional survey. Lancet. 2012; 379(9818): 815-822.
[CrossRef] [Google Scholar] [Pubmed]
- Galle J. Oxidative stress in chronic renal failure. Nephrol Dial Transplant. 2001; 16(11): 2135-2137.
[CrossRef] [Google Scholar] [Pubmed]
- Oberg BP, McMenamin E, Lucas FL, McMonagle E, Morrow J, Ikizler TA, et al. Increased prevalence of oxidant stress and inflammation in patients with moderate to severe chronic kidney disease. Kidney Int. 2004; 65(3): 1009-1016.
[CrossRef] [Google Scholar] [Pubmed]
- Yavuz O, Bicik Z, Cinar Y, Guney Y, Guler S. The effect of different dialysis membranes on oxidative stress and selenium status. Clin Chim Acta. 2004; 346(2): 153-160.
[CrossRef] [Google Scholar] [Pubmed]
- Locatelli F, Canaud B, Eckardt KU, Stenvinkel P, Wanner C, Zoccali C. Oxidative stress in end‐stage renal disease: An emerging threat to patient outcome. Nephrol Dial Transplant. 2003; 18(7): 1272-1280.
[CrossRef] [Google Scholar] [Pubmed]
- Sandstead HH. Trace elements in uremia and hemodialysis. Am J Clin Nutr. 1980; 33(7): 1501-1508.
[CrossRef] [Google Scholar] [Pubmed]
- Vanholder R, Cornelis R, Dhondt A, Lameire N. The role of trace elements in uraemic toxicity. Nephrol Dial Transplant. 2002; 17(2): 2-8.
[CrossRef] [Google Scholar] [Pubmed]
- Covic A, Gusbeth-Tatomir P. Trace elements in end-stage renal disease-unfamiliar territory to be revealed. BMC Nephrol. 2009; 10(1): 1-3.
[CrossRef] [Google Scholar] [Pubmed]
- Tonelli M, Wiebe N, Hemmelgarn B, Klarenbach S, Field C, Manns B, et al. Trace elements in hemodialysis patients: A systematic review and meta-analysis. BMC Med. 2009; 7(1): 1-2.
[CrossRef] [Google Scholar] [Pubmed]
- Ng AH, Hercz G, Kandel RE, Grynpas MD. Association between fluoride, magnesium, aluminum and bone quality in renal osteodystrophy. Bone. 2004; 34(1): 216-224.
[CrossRef] [Google Scholar] [Pubmed]
- Miyamoto T, Carrero JJ, Stenvinkel P. Inflammation as a risk factor and target for therapy in chronic kidney disease. Curr Opin Nephrol Hypertens. 2011; 20(6): 662-668.
[CrossRef] [Google Scholar] [Pubmed]
- Foley RN. Infectious complications in chronic dialysis patients. Perit Dial Int. 2008; 28(3): 167-171.
[CrossRef] [Google Scholar] [Pubmed]
- Collins AJ, Foley RN, Gilbertson DT, Chen SC. United States renal data system public health surveillance of chronic kidney disease and end-stage renal disease. Kidney Int Suppl. 2015; 5(1): 2-7.
[CrossRef] [Google Scholar] [Pubmed]
- Esfahani ST, Hamidian MR, Madani A, Ataei N, Mohseni P, Roudbari M, et al. Serum zinc and copper levels in children with chronic renal failure. Pediatr Nephrol. 2006; 21(8): 1153-1156.
[CrossRef] [Google Scholar] [Pubmed]
- Ranaldi G, Perozzi G, Truong-Tran A, Zalewski P, Murgia C. Intracellular distribution of labile Zn (II) and zinc transporter expression in kidney and Madin-Darby Canine Kidney (MDCK) cells. Am J Physiol Renal Physiol. 2002; 283(6): 1365-1375.
[CrossRef] [Google Scholar] [Pubmed]
- Shrimpton R, Gross R, Darnton-Hill I, Young M. Zinc deficiency: What are the most appropriate interventions? BMJ. 2005; 330(7487): 347-349.
[CrossRef] [Google Scholar] [Pubmed]
- Esfahani ST, Hamidian MR, Madani A, Ataei N, Mohseni P, Roudbari M, et al. Serum trace elements in children on maintenance hemodialysis. Acta Med Iran. 2016; 45(5): 351-354.
- Miura Y, Nakai K, Sera K, Sato M. Trace elements in sera from patients with renal disease. Nucl Instrum Methods Phys Res B. 1999; 150(1-4): 218-221.
- Krachler M, Wirnsberger GH. Long-term changes of plasma trace element concentrations in chronic hemodialysis patients. Blood Purif. 2000; 18(2): 138-143.
[CrossRef] [Google Scholar] [Pubmed]
- D'haese PC, de Broe ME. Adequacy of dialysis: Trace elements in dialysis fluids. Nephrol Dial Transplant. 1996; 11(2): 92-97.
[CrossRef] [Google Scholar] [Pubmed]
- Bhatnagar S, Natchu UC. Zinc in child health and disease. Indian J Pediatr. 2004; 71(11): 991-995.
[CrossRef] [Google Scholar] [Pubmed]
- Tzanno-Martins C, Azevedo LS, Orii N, Futata E, Jorgetti V, Marcondes M, et al. The role of experimental chronic renal failure and aluminium intoxication in cellular immune response. Nephrol Dial Transplant. 1996; 11(3): 474-480.
[CrossRef] [Google Scholar] [Pubmed]
- Sanaka T, Koremoto M. Selection guidelines for high-performance membrane. High-Performance Membrane Dialyzers. 2011; 173: 30-35.
[CrossRef] [Google Scholar] [Pubmed]
- Gokal R, Figueras M, Olle A, Rovira J, Badia X. Outcomes in peritoneal dialysis and haemodialysis: A comparative assessment of survival and quality of life. Nephrol Dial Transplant. 1999; 14(6): 24-30.
[CrossRef] [Google Scholar] [Pubmed]
- Vaziri ND, Oveisi F, Ding Y. Role of increased oxygen free radical activity in the pathogenesis of uremic hypertension. Kidney Int. 1998; 53(6): 1748-1754.
[CrossRef] [Google Scholar] [Pubmed]
- Himmelfarb J, Stenvinkel P, Ikizler TA, Hakim RM. The elephant in uremia: Oxidant stress as a unifying concept of cardiovascular disease in uremia. Kidney Int. 2002; 62(5): 1524-1538.
[CrossRef] [Google Scholar] [Pubmed]
- Deng G, Vaziri ND, Jabbari B, Ni Z, Yan XX. Increased tyrosine nitration of the brain in chronic renal insufficiency: Reversal by antioxidant therapy and angiotensin-converting enzyme inhibition. J Am Soc Nephrol. 2001; 12(9): 1892-1899.
[CrossRef] [Google Scholar] [Pubmed]
- Rükgauer M, Neugebauer RJ, Plecko T. The relation between selenium, zinc and copper concentration and the trace element dependent antioxidative status. J Trace Elem Med Biol. 2001; 15(2): 73-78.
[CrossRef] [Google Scholar] [Pubmed]
Author Info
Xiaojing Zhu1,2, Yuan Zhang1, Jin Han2, Dan Xiao2, Dan Zhu2 and Yan Ou2*2Department of Nephrology, The Second Affiliated Hospital of Xi’an Jiaotong University, Shaanxi, China
Citation: Zhu X: Effects of Chronic Kidney Disease on Levels of Oxidative Stress and Trace Elements
Received: 07-Feb-2022 Accepted: 22-Feb-2022 Published: 01-Mar-2022, DOI: 10.31858/0975-8453.13.3.128-134
Copyright: This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
ARTICLE TOOLS
- Dental Development between Assisted Reproductive Therapy (Art) and Natural Conceived Children: A Comparative Pilot Study Norzaiti Mohd Kenali, Naimah Hasanah Mohd Fathil, Norbasyirah Bohari, Ahmad Faisal Ismail, Roszaman Ramli SRP. 2020; 11(1): 01-06 » doi: 10.5530/srp.2020.1.01
- Psychometric properties of the World Health Organization Quality of life instrument, short form: Validity in the Vietnamese healthcare context Trung Quang Vo*, Bao Tran Thuy Tran, Ngan Thuy Nguyen, Tram ThiHuyen Nguyen, Thuy Phan Chung Tran SRP. 2020; 11(1): 14-22 » doi: 10.5530/srp.2019.1.3
- A Review of Pharmacoeconomics: the key to “Healthcare for All” Hasamnis AA, Patil SS, Shaik Imam, Narendiran K SRP. 2019; 10(1): s40-s42 » doi: 10.5530/srp.2019.1s.21
- Deuterium Depleted Water as an Adjuvant in Treatment of Cancer Anton Syroeshkin, Olga Levitskaya, Elena Uspenskaya, Tatiana Pleteneva, Daria Romaykina, Daria Ermakova SRP. 2019; 10(1): 112-117 » doi: 10.5530/srp.2019.1.19
- Dental Development between Assisted Reproductive Therapy (Art) and Natural Conceived Children: A Comparative Pilot Study Norzaiti Mohd Kenali, Naimah Hasanah Mohd Fathil, Norbasyirah Bohari, Ahmad Faisal Ismail, Roszaman Ramli SRP. 2020; 11(1): 01-06 » doi: 10.5530/srp.2020.1.01
- Manilkara zapota (L.) Royen Fruit Peel: A Phytochemical and Pharmacological Review Karle Pravin P, Dhawale Shashikant C SRP. 2019; 10(1): 11-14 » doi: 0.5530/srp.2019.1.2
- Pharmacognostic and Phytopharmacological Overview on Bombax ceiba Pankaj Haribhau Chaudhary, Mukund Ganeshrao Tawar SRP. 2019; 10(1): 20-25 » doi: 10.5530/srp.2019.1.4
- A Review of Pharmacoeconomics: the key to “Healthcare for All” Hasamnis AA, Patil SS, Shaik Imam, Narendiran K SRP. 2019; 10(1): s40-s42 » doi: 10.5530/srp.2019.1s.21
- A Prospective Review on Phyto-Pharmacological Aspects of Andrographis paniculata Govindraj Akilandeswari, Arumugam Vijaya Anand, Palanisamy Sampathkumar, Puthamohan Vinayaga Moorthi, Basavaraju Preethi SRP. 2019; 10(1): 15-19 » doi: 10.5530/srp.2019.1.3