Case Series - (2022) Volume 13, Issue 7
Abstract
Background: The prevalence of cardiovascular complications in COVID-19 infection varied in different studies. One of these complications is Myocardial Infarction. A disturbance of the blood supply can lead to Myocardial Infarction by clot formation in the arteries. However, no evidence of significant coronary stenosis has been found in more than 50% of patients with COVID-19 and ST elevation.
Case presentation: 38 and 49 years old men (patients 1, 2) were admitted to our hospital with the complaint of typical chest pain and COVID-19 symptoms. The Real-time Polymerase Chain Reaction (RT-PCR) test confirmed COVID-19 in both. Patient 1 represented inferior posterior ST-Elevation Myocardial Infarction (STEMI) in his Electrocardiogram (ECG). Also, patient 2 has ST-elevation in high lateral and septal leads (I, Augmented Vector Left (AVL), V1, V2) and ST-segment depression in AVR and inferior leads (III, Augmented Vector Foot (AVF)). Their troponin was positive. The vital signs were normal in both of them. Patient 2 just had a history of Aortic Valve Replacement (AVR) 5 years ago. However, Patient 1 had no medical history. Trans-Thoracic Echocardiography (TTE) data demonstrated some disturbances in patient 1 severe hypokinesia of inferior, posterior, lateral, and septal walls. However, TTE data were unremarkable for patient 2. We prescribed recommended medications for them. Therefore, their ECG changes were corrected, and his condition improved. In addition, coronary angiography was done that demonstrated patent and normal coronary arteries in both of them.
Conclusion: COVID-19 infection can cause normal coronary arteries Myocardial Infarction with probable two mechanisms prolonged vasospasm or intraluminal coronary thrombogenesis.
Keywords
ST-elevation, Normal coronary, Myocardial Infarction (MI), COVID-19, ST-Elevation Myocardial Infarction (STEMI)
Abbreviations
COVID-19: Corona Virus Disease 2019; ACS: Acute Coronary Syndrome; ECG: Electrocardiogram; TTE: Trans-Thoracic Echocardiography; MI: Myocardial Infarction; STEMI: ST-Elevation Myocardial Infarction
Introduction
From the beginning of the COVID-19 epidemic in December 2019, more than 15 million people got infected by COVID-19 until 26 July 2020 (WHO, 2020). COVID-19 pandemic causes a significant burden on medical institutions. In such a pandemic, the management of emergencies like Myocardial Infarction (MI) was too complicated (Kim HN, etal., 2020). Cardiac risk factors expose the patients to more complex or clinical situations that require the Intensive Care Unit (ICU) (Huang C, et al., 2020). Myocardial injury due to ischemia, acute thrombotic obstruction, or myocarditis has been reported in approximately 7% to 28% of hospitalized patients with COVID-19. Studies have shown that these injuries are associated with increased mortality (Guo T, etal., 2020; Shi S, etal., 2020; Wang D, etal., 2020). Traditionally, ST-Elevation Myocardial Infarction (STEMI) is the consequence of coronary artery stenosis. It means we can find some coronary arteries with an abnormal situation. In these cases, we reported two patients with COVID-19 infection presented with STEMI and the normal coronary artery.
Case Presentation
Case 1
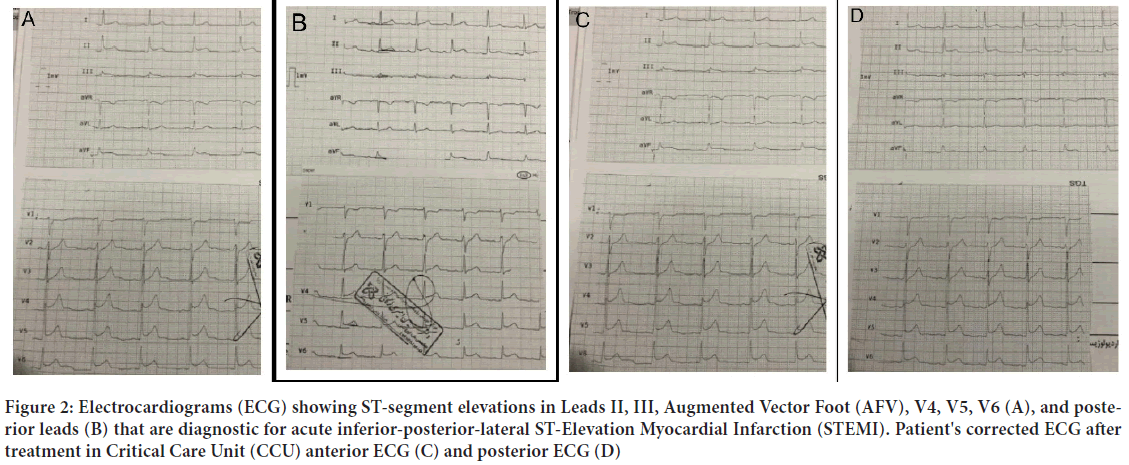
A 38-years-old man complained of pain in the neck, chest, and right hand two days after taking azithromycin. He also had symptoms of COVID-19 such as fever, sore throat, weakness, and myalgia. As the pain intensified, he went to the hospital's emergency ward. The patient felt shortness of breath during the visit. However, he had no fever, chills, cough, or sputum, and a normal heart rate. As a result, Real-Time PCR was performed on him, and he was identified as a COVID-19 patient. His treatment was started according to the COVID-19 treatment protocol. The patient reported no history of cardiovascular and pulmonary diseases, thromboembolism, or coagulation. Also, he did not take any medication. On his arrival, physical examinations were performed; his O2 saturation was 100%, blood pressure was 133/88 mmHg, and pulse rate was 85 beats per minute. The Electrocardiography (ECG) showed inferior-posterior STEMI in inferior ECG view and posterior ECG (Figure 1). The laboratory results are shown in Table 1. Trans-Thoracic Echocardiography (TTE) study demonstrated severe hypokinesia of inferior, posterior, lateral, and septal LV (Left Ventricle) walls with mild mitral, tricuspid, and pulmonary valves regurgitation. LV ejection fraction and systolic pulmonary pressure were estimated at 40% and 20 mmHg, respectively. Other acquired data from TTE were normal. Therefore, we hospitalized him in Critical Care Unit (CCU) with a diagnosis of COVID-19 and acute inferior-posterolateral MI. Treatment was begun including Atorvastatin, Aspirin, Plavix, Pantoprazole, Enoxaparin, Reteplase, and Nitroglycerin (TNG) with recommended doses in standard guidelines. His condition improved with these treatments, and ECG changes were corrected in anterior ECG and posterior ECG (Figure 1). He underwent coronary angiography for rescue PCI (Percutaneous Coronary Intervention). The next day, angiography was performed for him, showing normal coronary vessels without any Intimal irregularity, atherosclerosis plaque, and thrombosis (Figure 2). The patient was discharged with a good general condition and a normal examination.
| Laboratory findings | Result | Normal range |
|---|---|---|
| FBS (Fasting Blood Sugar) | 182 | Adult (>20 years): 70-115 |
| CPK | 686 u/l | Male: 24-195 u/l |
| Female: 24-170 u/l | ||
| CPK-MB | 86 u/l | <24 u/l |
| Troponin I (1st time) | Positive weakly | |
| Troponin I (2nd time) | Positive | |
| CPR | ++ | |
| PT | 13 | 10.5-13.5 |
| PTT | 38 | 26-40 |
Note: FBS: Fasting Blood Sugar; CPK: Creatine phosphokinase; CPK-MB: Creatine Phosphokinase-MB; CRP: C-Reactive Protein; PT: Prothrombin Time; PTT: Partial Thromboplastin Time
Table 1: The results of serology laboratory assays done during CCU (Coronary Care Unit) admission
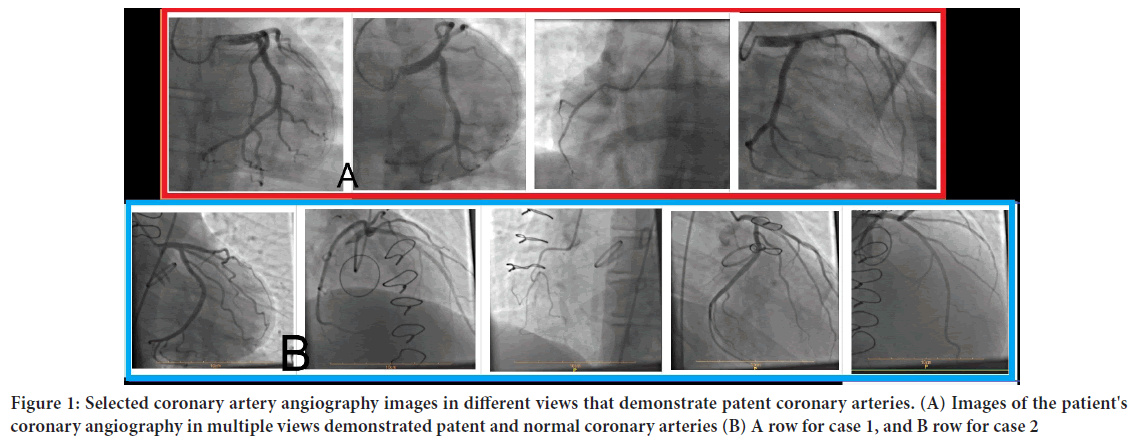
Figure 1: Selected coronary artery angiography images in different views that demonstrate patent coronary arteries. (A) Images of the patient's coronary angiography in multiple views demonstrated patent and normal coronary arteries (B) A row for case 1, and B row for case 2
Figure 2: Electrocardiograms (ECG) showing ST-segment elevations in Leads II, III, Augmented Vector Foot (AFV), V4, V5, V6 (A), and posterior leads (B) that are diagnostic for acute inferior-posterior-lateral ST-Elevation Myocardial Infarction (STEMI). Patient's corrected ECG after treatment in Critical Care Unit (CCU) anterior ECG (C) and posterior ECG (D)
Case 2
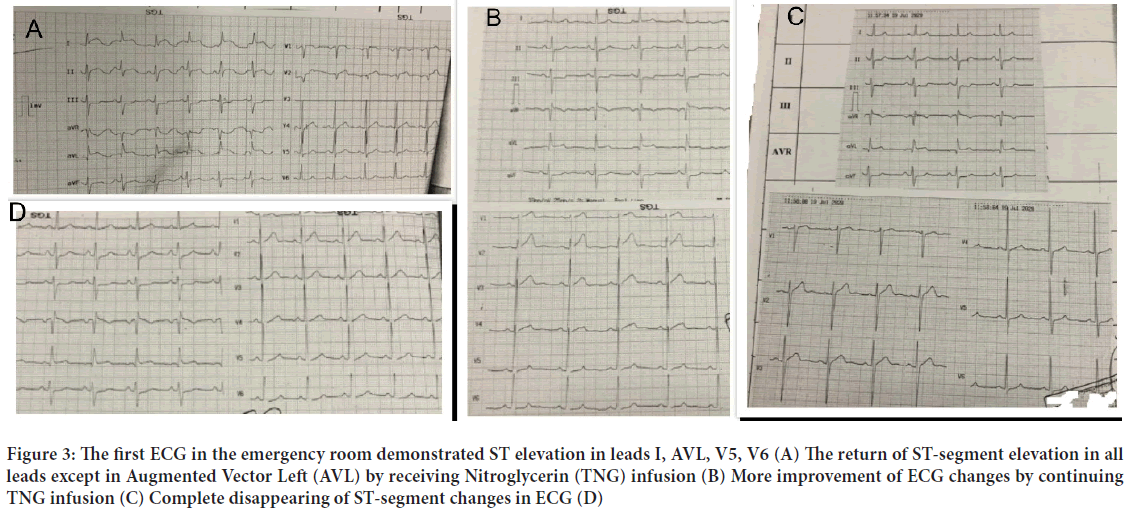
A 49-year-old man was recently admitted to our hospital with the chief complaint of retrosternal typical chest pain, which released to the back and both hands and had started from 30 minutes before admission with a slowly increasing trend. He also had a headache, myalgia and lethargy, cough, dyspnea, and anorexia 48 hours ago, which can be the COVID-19 infection symptoms. He had a history of Aortic Valve Replacement (AVR) 5 years ago with good monitoring of INR (International Normalized Ratio). His blood pressure was 135/95 mmHg, and his heart rate was 85 regular beats per minute. O saturation was 96% without O therapy in room air. In the physical exam, he was aware and awake with good obey. The metallic valve sound was heard well. No rales and wheeze were heard in the lung exam. Spiral chest CT-scan demonstrated widespread bilateral lesions in favor of COVID-19 pneumonia. The first Electrocardiogram in the emergency room revealed sharp and typical ST-segment elevation in high lateral and septal leads (I, AVL, V1, V2) together with ST-segment depression in AVR and inferior leads (III, AVF) in favor of acute ischemia (Figure 3).
Figure 3: The first ECG in the emergency room demonstrated ST elevation in leads I, AVL, V5, V6 (A) The return of ST-segment elevation in all leads except in Augmented Vector Left (AVL) by receiving Nitroglycerin (TNG) infusion (B) More improvement of ECG changes by continuing TNG infusion (C) Complete disappearing of ST-segment changes in ECG (D)
TTE showed mild hypokinesia in the lateral wall with mild mitral regurgitation and good prosthetic aortic valve function. The first Complete blood count (CBC) showed leukopenia (White Blood Cells (WBC): 3500) with normal difference. His INR was 2, and C-reactive protein (CRP) was 2 mg/L (Table 2). Immediately, we began anti-ischemic therapy containing ASA (Aspirin) 300 mg, Plavix 300 mg, atorvastatin 80 mg stat, and TNG drip 5 µg/min. Due to ST-segment elevation, the patient rapidly transferred to COVID-19 positive CCU for further evaluation and treatment of ECG changes, management of COVID-19, and maybe primary PCI. The first qualified troponin in the emergency room and the second 6 hours later was negative. In the second ECG after 20 minutes, TNG infusion chest pain was subsided almost entirely, and all ST elevation returned to baseline except in AVL (Figure 3). Over time and with receiving TNG infusion, continued improvement course of symptoms and ECG changes resolving (Figure 3). After 2 hours, all ECG changes were corrected completely, and the patient was without any chest pain (Figure 3). The third qualified troponin was weekly positive. Two days later, coronary angiography demonstrated complete patent and completely normal epicardial coronary arteries (Figure 2). The patient was discharged after treatment of COVID-19 and improvement of other symptoms and signs with a calcium blocker, beta-blocker, and Warfarin.
| Laboratory findings | Result | Normal range |
|---|---|---|
| FBS (blood sugar) | 102 | Adult (>20 years): 70-115 |
| CPK | 160 u/l | Male: 24-195 u/l |
| Female: 24-170 u/l | ||
| CPK-MB | 26 u/l | <24 u/l |
| Troponin I (1st time) | negative | Negative |
| Troponin I (2nd time) | negative | Negative |
| Troponin I (3rd time) | weekly positive | Negative |
| CPR | 2 mg/l | ≤ 1 mg/l |
| INR | 2 IU | ≤ 1.5 IU |
| CBC | 3500/mm3 | 3500-10000/mm3 |
| LDH | 724 IU/L | 313-618 IU/L |
| PLT | 136000/mm3 | 150000-450000/mm3 |
Note: FBS: Fasting Blood Sugar; CPK: Creatine phosphokinase; CPK-MB: Creatine phosphokinase-MB; CRP: C-Reactive Protein; INR: International Normalized Ratio; CBC: Complete Blood Count; LDH: Lactate Dehydrogenase; PLT: Platelet
Table 2: Laboratory findings measured during admission
Discussion
Cardiovascular complications have been reported in some COVID-19 cases. Frequently, myocardial injury has been seen between them (Ong E, et al., 2021). The prevalence of cardiovascular disease among COVID-19 patients varies from 4% to 14.6%. Therefore, it remained uncertain whether hypertension, diabetes mellitus, and cardiovascular disease are related to COVID-19 disease severity (Kim HN, etal., 2020). Some studies reported COVID-19 cases with cardiac injuries such as pericarditis, myocarditis, arrhythmia, STEMI, and coronary artery thrombosis (Dabbagh MF, etal., 2020; Monmeneu JV, etal., 2020; Dominguez-Erquicia P, etal., 2020; Stefanini GG, etal., 2020). The prevalence of Myocardial Infarction between women and men is 30% and 12%, respectively. However, it was seen that in more than 50% of MI, there isn't any evidence of obstructive coronary disease at the time of angiography (Reynolds HR, et al., 2011; Gehrie ER, et al., 2009). There are some reasons for Myocardial Infarction with nonobstructive coronary arteries:
•Prolonged coronary spasm can occlude the coronary artery and lead to Myocardial Infarction.
•Coronary spam activates platelet and leads to acute coagulation process (Agewall S, etal., 2017; Nakayama N, etal., 2014).
•Releasing emboli from enlarged and thrombotic cardiac chambers and transfer into coronary arteries.
•Some risk factors develop coronary vasospasm like cocaine, tobacco, and nicotine (Gharacholou SM, etal., 2017). Moderate to severe valvular heart disease and atrial fibrillation/flutter rhythm are important risk factors for coronary emboli. In this case that we presented, the patient had no history of atherothrombotic disorders or significant cardiovascular diseases, and his cardiac all chamber size was normal. In his ECG, acute inferior-posterior-lateral STEMI was observed after treatment and angiography. Atherosclerosis plaque or intimal irregularity in the coronary arteries was not seen. According to our findings, there are two possibilities for the explanation of this event:
•Some studies have reported that COVID-19 causes significant inflammation and coagulation, which is activated by an inflammatory response (Connors JM and Levy JH, 2020). In this patient, inflammatory factors were higher than normal; therefore, COVID-19 probably causes thrombus in the coronary artery and leads to acute inferior-posterior-lateral MI. Then, the patient use Reteplase to resolve this thrombus. Consequently, changes of ECG were corrected, and we didn't see any thrombus in angiography.
•Our patient didn't use any drugs that cause prolonged spasms; therefore, COVID-19 probably leads to prolonged spasms in the coronary artery and, finally acute MI.
Conclusion
Regardless of Myocardial Infarction pathogenesis, we must note that COVID-19 infection can cause ST-elevation and MI, even in cases with normal coronary arteries and without any atherosclerosis plaque. COVID-19 infection can cause normal coronary arteries Myocardial Infarction with probable two mechanisms prolonged vasospasm or intraluminal coronary thrombogenesis. It appears that infection with COVID-19 can cause prolonged coronary artery spasm or clot formation in the arteries, which can eventually lead to Myocardial Infarction.
Declaration
This study complies with the Declaration of Helsinki that the locally appointed ethics committee has approved the research protocol and that the subjects have obtained informed consent.
References
- WHO. Coronavirus Disease (COVID-19): Situation Report. World Health Organization. 2020.
- Kim HN, Lee JH, Park HS, Yang DH, Jang SY, Bae MH, et al. A case of COVID-19 with acute myocardial infarction and cardiogenic shock. J Korean Med Sci. 2020; 35(27).
[Crossref] [Google Scholar] [Pubmed]
- Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020; 395(10223): 497-506.
[Crossref] [Google Scholar] [Pubmed]
- Guo T, Fan Y, Chen M, Wu X, Zhang L, He T, et al. Cardiovascular implications of fatal outcomes of patients with coronavirus disease 2019 (COVID-19). JAMA Cardiol. 2020; 5(7): 811-818.
[Crossref] [Google Scholar] [Pubmed]
- Shi S, Qin M, Shen B, Cai Y, Liu T, Yang F, et al. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan, China. JAMA Cardiol. 2020; 5(7): 802-810.
[Crossref] [Google Scholar] [Pubmed]
- Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020; 323(11): 1061-1069.
[Crossref] [Google Scholar] [Pubmed]
- Ong E, Castro‐Dominguez Y, Brennan J, Oen‐Hsiao J. COVID‐19 complicated by St‐Segment Elevation Myocardial Infarction in a 29‐year‐old patient. Catheter Cardiovasc Interv. 2021; 97(2): 267-271.
[Crossref] [Google Scholar] [Pubmed]
- Dabbagh MF, Aurora L, D’Souza P, Weinmann AJ, Bhargava P, Basir MB. Cardiac tamponade secondary to COVID-19. JACC Case Rep. 2020; 2(9): 1326-1330.
[Crossref] [Google Scholar] [Pubmed]
- Monmeneu JV, Mafe ED, Soler JA, Perez BV, Caravaca JS, Torres RB, et al. Subacute perimyocarditis in a young patient with COVID-19 infection. Eur Heart J Case Rep. 2020.
[Crossref] [Google Scholar] [Pubmed]
- Dominguez-Erquicia P, Dobarro D, Raposeiras-Roubín S, Bastos-Fernandez G, Iñiguez-Romo A. Multivessel coronary thrombosis in a patient with COVID-19 pneumonia. Eur Heart J. 2020; 41(22): 2132.
[Crossref] [Google Scholar] [Pubmed]
- Stefanini GG, Montorfano M, Trabattoni D, Andreini D, Ferrante G, Ancona M, et al. ST-elevation myocardial infarction in patients with COVID-19: Clinical and angiographic outcomes. Circulation. 2020; 141(25): 2113-2116.
[Crossref] [Google Scholar] [Pubmed]
- Reynolds HR, Srichai MB, Iqbal SN, Slater JN, Mancini GJ, Feit F, et al. Mechanisms of myocardial infarction in women without angiographically obstructive coronary artery disease. Circulation. 2011; 124(13): 1414-1425.
[Crossref] [Google Scholar] [Pubmed]
- Gehrie ER, Reynolds HR, Chen AY, Neelon BH, Roe MT, Gibler WB, et al. Characterization and outcomes of women and men with non-ST-segment elevation myocardial infarction and nonobstructive coronary artery disease: Results from the Can Rapid risk stratification of Unstable Angina patients suppress adverse outcomes with Early implementation of the ACC/AHA Guidelines (CRUSADE) quality improvement initiative. Am Heart J. 2009; 158(4): 688-694.
[Crossref] [Google Scholar] [Pubmed]
- Agewall S, Beltrame JF, Reynolds HR, Niessner A, Rosano G, Caforio AL, et al. ESC working group position paper on myocardial infarction with non-obstructive coronary arteries. Eur Heart J. 2017; 38(3): 143-153.
[Crossref] [Google Scholar] [Pubmed]
- Nakayama N, Kaikita K, Fukunaga T, Matsuzawa Y, Sato K, Horio E, et al. Clinical features and prognosis of patients with coronary spasm-induced non-ST‐segment elevation acute coronary syndrome. J Am Heart Assoc. 2014; 3(3): 795.
[Crossref] [Google Scholar] [Pubmed]
- Gharacholou SM, Ijioma N, Banwart E, Munoz FD. ST-segment elevation myocardial infarction and normal coronary arteries after consuming energy drinks. Case Rep Cardiol. 2017.
[Crossref] [Google Scholar] [Pubmed]
- Connors JM, Levy JH. COVID-19 and its implications for thrombosis and anticoagulation. Blood. 2020; 135(23): 2033-2040.
[Crossref] [Google Scholar] [Pubmed]
Author Info
Erfan Kazemi1, Mehran Gheshlaghi1, Ali Mansoursamaei1, Saba Homatash2 and Hossein Sheibani2*2Department of Clinical Research Development Unit, Imam Hossein Hospital, Shahroud University of Medical Science, Shahroud, Iran
Citation: Kazemi E: Normal Coronary Myocardial Infarction Due to COVID-19: A Case Series
Received: 30-Jun-2022 Accepted: 22-Jul-2022 Published: 29-Jul-2022, DOI: 10.31858/0975-8453.13.7.483-486
Copyright: This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
ARTICLE TOOLS
- Dental Development between Assisted Reproductive Therapy (Art) and Natural Conceived Children: A Comparative Pilot Study Norzaiti Mohd Kenali, Naimah Hasanah Mohd Fathil, Norbasyirah Bohari, Ahmad Faisal Ismail, Roszaman Ramli SRP. 2020; 11(1): 01-06 » doi: 10.5530/srp.2020.1.01
- Psychometric properties of the World Health Organization Quality of life instrument, short form: Validity in the Vietnamese healthcare context Trung Quang Vo*, Bao Tran Thuy Tran, Ngan Thuy Nguyen, Tram ThiHuyen Nguyen, Thuy Phan Chung Tran SRP. 2020; 11(1): 14-22 » doi: 10.5530/srp.2019.1.3
- A Review of Pharmacoeconomics: the key to “Healthcare for All” Hasamnis AA, Patil SS, Shaik Imam, Narendiran K SRP. 2019; 10(1): s40-s42 » doi: 10.5530/srp.2019.1s.21
- Deuterium Depleted Water as an Adjuvant in Treatment of Cancer Anton Syroeshkin, Olga Levitskaya, Elena Uspenskaya, Tatiana Pleteneva, Daria Romaykina, Daria Ermakova SRP. 2019; 10(1): 112-117 » doi: 10.5530/srp.2019.1.19
- Dental Development between Assisted Reproductive Therapy (Art) and Natural Conceived Children: A Comparative Pilot Study Norzaiti Mohd Kenali, Naimah Hasanah Mohd Fathil, Norbasyirah Bohari, Ahmad Faisal Ismail, Roszaman Ramli SRP. 2020; 11(1): 01-06 » doi: 10.5530/srp.2020.1.01
- Manilkara zapota (L.) Royen Fruit Peel: A Phytochemical and Pharmacological Review Karle Pravin P, Dhawale Shashikant C SRP. 2019; 10(1): 11-14 » doi: 0.5530/srp.2019.1.2
- Pharmacognostic and Phytopharmacological Overview on Bombax ceiba Pankaj Haribhau Chaudhary, Mukund Ganeshrao Tawar SRP. 2019; 10(1): 20-25 » doi: 10.5530/srp.2019.1.4
- A Review of Pharmacoeconomics: the key to “Healthcare for All” Hasamnis AA, Patil SS, Shaik Imam, Narendiran K SRP. 2019; 10(1): s40-s42 » doi: 10.5530/srp.2019.1s.21
- A Prospective Review on Phyto-Pharmacological Aspects of Andrographis paniculata Govindraj Akilandeswari, Arumugam Vijaya Anand, Palanisamy Sampathkumar, Puthamohan Vinayaga Moorthi, Basavaraju Preethi SRP. 2019; 10(1): 15-19 » doi: 10.5530/srp.2019.1.3